As an Indian food manufacturer or a dynamic food business operator, you are well aware of the huge potential that the international market offers, especially the discerning consumers of the United States of America. However, it is important to note that while quality products are a prerequisite for entering this market, it is also important to comply with stringent international standards. In this regard, your USFDA food products certification is your passport to this market. It is not merely a formality; it is a reflection of your commitment to quality and safety, which enables your delicious food products to reach the tables of discerning consumers in the USA.
In this blog post, we intend to simplify the USFDA food products certification process for you.
Talk to Our Experts Today
Join 500+ Indian Food Exporters Who Got USFDA Certified with Legal4sure. Book a Free 1 Hour Consultation with Legal4sure’s FDA Expert — We Handle Registration, Labeling, Prior Notice and FSVP. Call Legal4sure Today.
Why Businesses Struggle With USFDA Food Registration
However, many ambitious Indian food businesses are at a crossroads while targeting the highly profitable US market. Despite producing high-quality food products, these businesses are likely to face major hurdles. The major problem is related to international regulations, resulting in delays, rejections, and sometimes an inability to export. Food products without USFDA certification are likely to be stuck in ports, resulting in high demurrage costs or being refused entry into the US.
The Root Causes
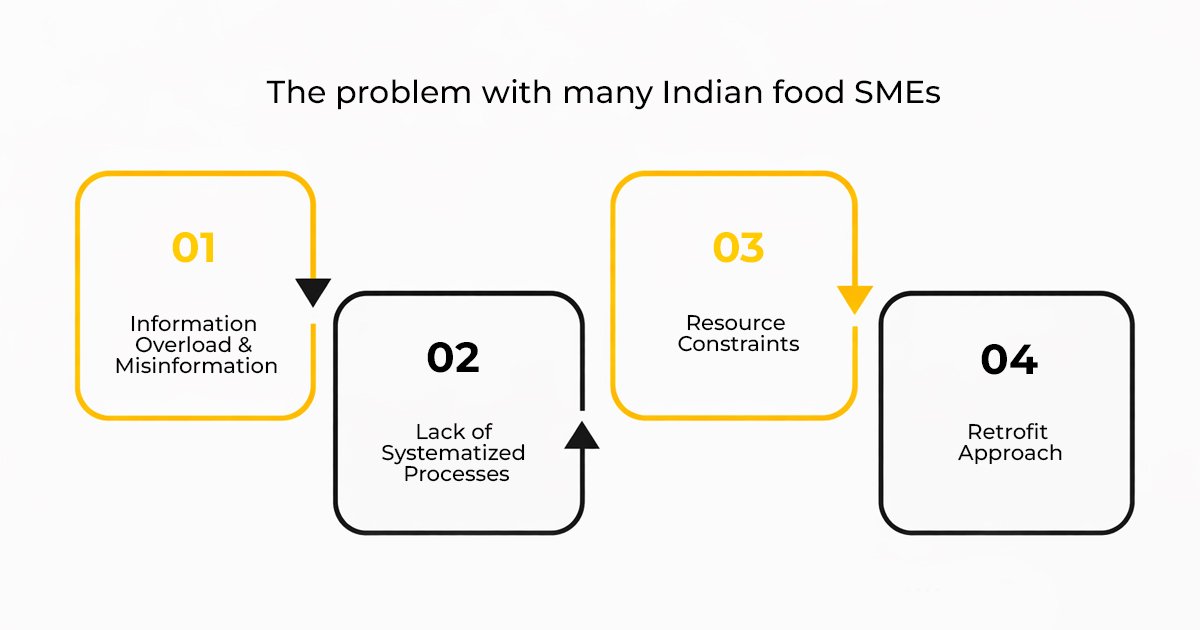
The problem with many Indian food SMEs is not that they are not trying hard enough to export to the US market; the problem is a list of gaps:
- Information Overload & Misinformation: There is a lot of information on USFDA regulations, which may be intimidating for food businesses. There is a gap between actual and perceived barriers.
- Lack of Systematized Processes: There is a lack of systematized processes in quality control and documentation. This is a critical gap because fda food registration requires systematized processes.
- Resource Constraints: There are resource constraints because food businesses are not able to afford separate teams for USFDA compliance and expert consultation on the intricate legal issues.
- Retrofit Approach: There is a lack of systematized processes because food businesses are trying to retrofit existing processes to USFDA regulations at the last moment.
>Helpful Guide:- What is USFDA Certification and Why It Matters
Answering Your USFDA Queries
As an entrepreneur or business leader, you probably have burning questions about the USFDA food export process. Here are some of the questions that might be on your mind:
- Is the USFDA food product certification a one-time procedure or an ongoing process?
- What will it really cost my small business, and will the ROI be worth it?
- Will the fda food registration procedure cause any interruption in my production schedules?
- Do I need to hire consultants to assist in the procedure, or is it something that can be done in-house?
- Will it really get my foot in the door of the big US retail accounts and international clients, or is it just more paperwork?
USFDA Food Certification Requirements
The United States Food and Drug Administration (USFDA) maintains high standards in ensuring the safety of food consumed in the United States. It is a mandate for any foreign food facility intending to export foodstuffs to the United States of America to comply with these regulations. The fundamental regulations encompass two major areas:
- Facility Registration: All foreign food facilities intending to manufacture, process, pack, or hold food for consumption in the United States must register with the FDA. This falls under the fda food registration regulations under the Federal Food, Drug, and Cosmetic (FD&C) Act.
- Adherence to FSMA (Food Safety Modernization Act): This legislation provides a paradigm shift in food safety regulations from responding to food contamination to preventing it. Major components of the legislation include:
- Preventive Controls: A food safety plan with hazard analysis and risk-based preventive controls must be implemented.
- Supplier Verification: Importers must verify foreign suppliers’ compliance with United States food safety regulations.
- Foreign Supplier Verification Programs (FSVP): United States importers of foodstuffs must implement these programs in line with food imported into the United States.
- Record-Keeping: Thorough documentation of processes and controls is essential in the process of obtaining fda food approval.
>Helpful Guide:- Documents Required For USFDA Certification In India 2026 | Complete Checklist
USFDA Food Certification Process
While attaining USFDA food approval is a challenging task, it is a well defined process that, if completed accurately, will result in a successful outcome. Here is a simple step by step guide to attaining USFDA food certification:
- Go Through The FSMA Requirements: Familiarize yourself with the requirements under FSMA that are applicable to your type of food product. This is generally related to Preventive Controls for Human Food.
- Appoint a US Agent: Every foreign facility must appoint a US Agent to act as a liaison with FDA. This is a critical component of USFDA food certification.
- Facility Registration: Register your food processing facility with FDA. This must be done every two years (biennially) between October 1st and December 31st in even-numbered years.
- Develop a Food Safety Plan: Develop a detailed food safety plan based on HACCP (Hazard Analysis and Critical Control Points) principles and FSMA’s Preventive Controls requirements.
- Labeling Compliance: Make sure that your product’s labeling is compliant with FDA’s stringent guidelines for ingredients, nutritional information, allergen statements, and other mandatory disclosures.
- Maintain Records: Keep detailed records of all activities related to production, sanitation procedures, training activities, and food safety monitoring. These records are essential during inspections.
- Importer Collaboration (FSVP): If you are exporting your product, your US importer must establish and maintain a Foreign Supplier Verification Program (FSVP) to verify that your product meets US food safety standards. Be ready to provide your US importer with necessary documentation.
- Prepare for Inspections: The FDA has the authority to inspect your facility if it is a foreign food facility. Ensure that your facility is audit ready at all times by having robust food safety procedures in place.
Key Benefits of USFDA Certification
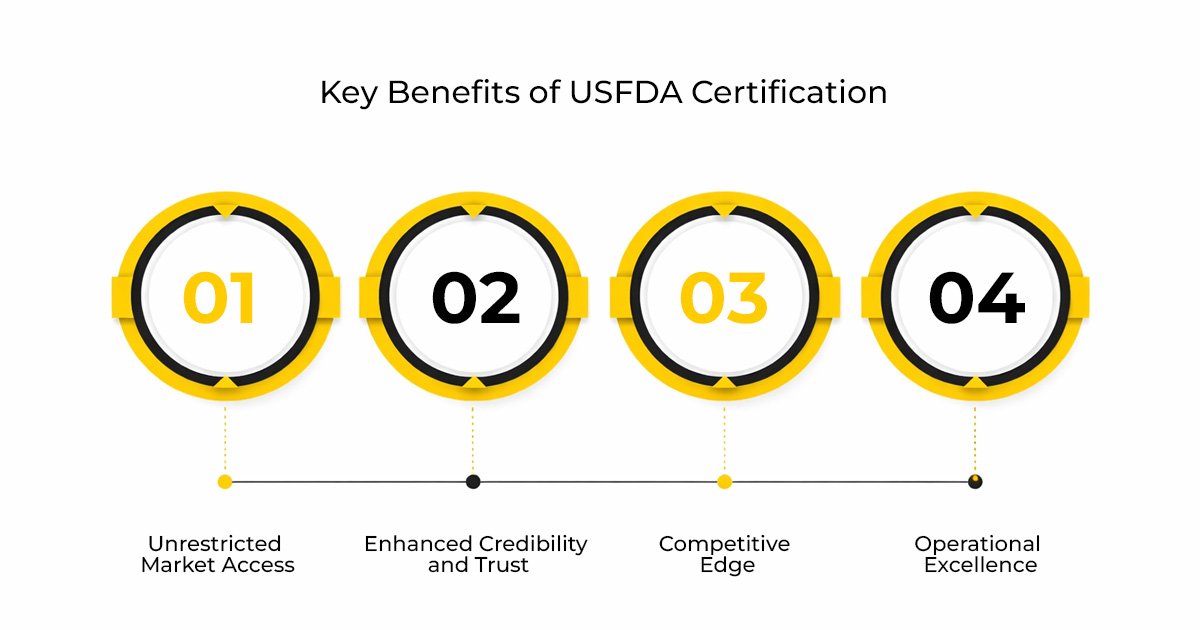
Obtaining USFDA certification for your food products has many advantages for Indian food businesses:
- Unrestricted Market Access: You can begin your usfda food export process and it will act as a major boost to your business as you will get direct entry to the huge and profitable US food market.
- Enhanced Credibility and Trust: With USFDA food certification, you can establish immediate confidence with US food importers, distributors, and consumers about your brand and its commitment to quality.
- Competitive Edge: Stand out from your peers who are yet to embark on the USFDA food certification journey and give your business a competitive advantage over your competition.
- Operational Excellence: The USFDA food certification process itself is a transformative experience that can lead to operational efficiency and a more streamlined business environment.
>Helpful Guide:- How to Get US FDA Certification in India in a Simple Way
Key Takeaways for Founders
- Don’t wait until you get your export order to start worrying about USFDA. Start worrying about USFDA from Day One itself as part of your business plan.
- While self-learning is good, a seasoned consultant can save you immense time and prevent costly mistakes. His or her advice is invaluable.
- Meticulous documentation is your greatest strength in any audit or verification situation.
- Food safety is every individual’s responsibility. Training and a culture of compliance are non-negotiables.
- Having a good relationship with an importer who is well-informed about FSVP is critical to your exports.
>Helpful Guide:- USFDA Certification Cost 2026 – Full Fee Breakdown & What You Must Know
Frequently Asked Questions
What is the usfda certification for food export from india process?
The steps include facility registration, the appointment of a US agent, the development of an effective Food Safety Plan, compliance with labeling requirements, and record maintenance.
What are the fda approval requirements for food products india?
In the case of food products, the main requirements include facility registration, compliance with the FSMA’s preventive controls and accurate labeling.
What is the usfda food facility registration process india?
Foreign food facilities must register with the FDA using an online system and must include details about their facility and the appointment of a US Agent.
Does pharma need fda approval?
Pharmaceutical drugs and devices involve the FDA approval procedures that include clinical trials and the submission of data, which is different from the case of food products.
What are the usfda pharma requirements?
The major requirements include the investigational new drug application compliance with Good Manufacturing Practices (GMP), and post-market surveillance.
What is the fda pharma process?
The FDA pharma approval process includes pre-clinical research, clinical trials (Phase I, Phase II, and Phase III), the submission of the New Drug Application (NDA), and the post-market surveillance.
How often must I renew my FDA Food Registration?
Every two years. Renewals must be submitted between October 1 and December 31 of every even-numbered year (e.g., 2026).