For food manufacturers and exporters in India who have aspirations to break into the lucrative U.S. food industry, gaining USFDA approval is not just a requirement, it is a ticket to huge growth potential. However, to gain approval, it seems like a journey through a maze of complexities, especially regarding understanding the actual USFDA registration fees involved. Many food manufacturers and exporters in India are plagued by the uncertainty of expenses, charges, and efforts involved in complying with international regulations. This blog aims to simplify the concept of FDA registration cost India 2026, helping readers understand exactly what it entails in terms of expenses, uncovering potential hidden charges, and ultimately revealing to them exactly what it will cost to gain USFDA Food Facility Registration approval.
Talk to Our Experts Today
Not sure how much USFDA food facility registration actually costs in India? Legal4Sure experts break down every fee — official, hidden & total — so you pay nothing unexpected. Get a free personalised estimate from Legal4Sure today.
Frequent Questions of Founders About USFDA Registration Fees
For the founder of the business, the decision regarding the registration with the USFDA comes with a flood of questions, including the following:
- Are the fees for the registration with the USFDA fixed, or are they variable depending on the nature of the food product?
- Are there going to be any hidden costs that are not included in the original quotation for the FDA registration cost India?
- How can I ensure that the fees quoted by the consultant for the FDA price India are reasonable and competitive?
- What is the real impact on the budget in terms of the fees for the initial registration, as well as the maintenance fees on an annual basis and the renewal fees on a bi-annual basis?
- Can a small business really afford the entire investment required for the compliance with the USFDA, including the USFDA fees India?
>Helpful Guide:- What is USFDA Certification and Why It Matters
Simplifying Your USFDA Registration Fees
For food business operators in India, it is a crossroads when it comes to USFDA registration. The main problem faced by food business operators in India is the high fees charged by the USFDA for registration and a lack of transparency in this regard. Food business operators in India often face a problem in planning a budget for a business in which there is a high probability that the cost of FDA registration in India will be more than initially anticipated by food business operators in India. This acts as a major hindrance for food business operators in India, causing them to miss opportunities in a world that is eager for quality food products from India.
Even though there is no fee for registration in the case of food facility registration in the USFDA, it is important to know that there are various components in determining the entire USFDA registration fees:
|
Category |
Official USFDA Fee (FY 2025/26) |
Approx. Cost in India (INR) |
| USFDA Registration Fee (Direct) | $0 (Free) | ₹0 |
| FDA User Fees (Re-inspection) | $339 – $376 per hour | ₹28,500 – ₹32,000 / hr |
| Agent Fees (Mandatory) | Managed by Private Bodies | ₹30,000 – ₹65,000 / yr |
| Consultancy Fees (Full Service) | Managed by Private Bodies | ₹15,000 – ₹45,000 |
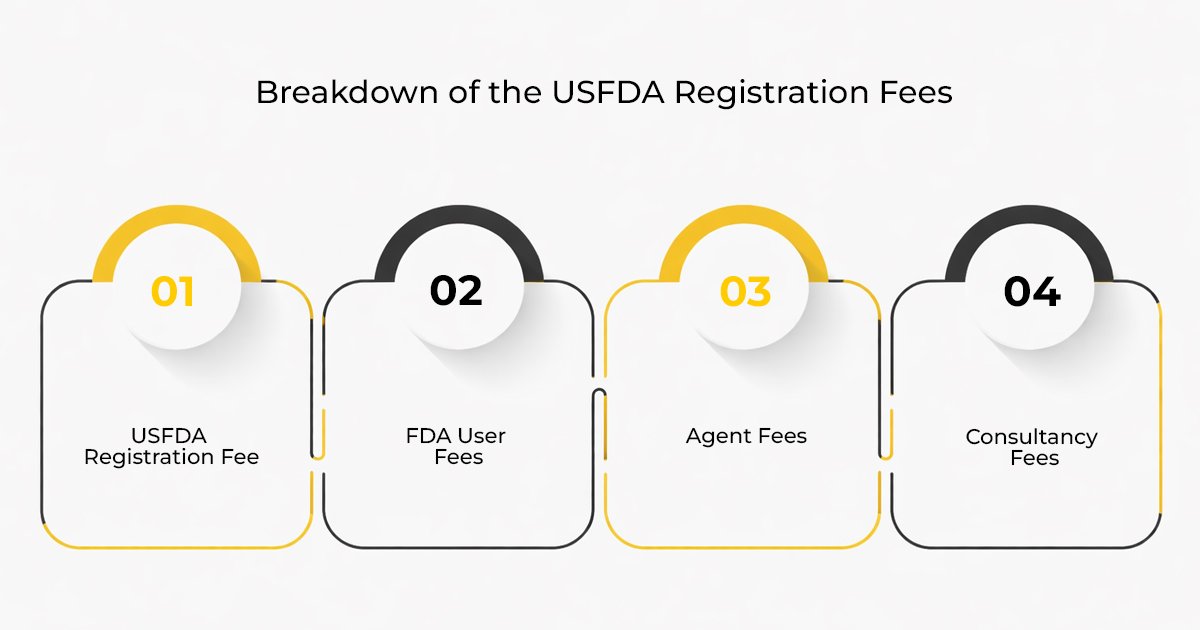
Breakdown of the USFDA Registration Fees
- USFDA Registration Fee
Currently, as of 2026, there is no direct registration fee with the USFDA. The registration is for free if you register directly through their online portal (FIS). The mention of a Government Fee by a consultant for basic food registration is a red flag.
- FDA User Fees
Registration is free of charge, but there may be a charge for the user. For example, if your facility is under an FDA inspection, there may be a fee for the inspection. This may be applicable in cases like re-inspection of the facility or if the facility is part of a specialized program like VQIP.
Note: The foreign reinspection fee is approximately $376 (approx. ₹35,000) per hour, excluding the inspector’s travel and lodging costs, as of FY 2026.
- Agent Fees
For facilities outside of the U.S., a U.S. Agent must be appointed. This is a person who is physically present in the U.S. and is authorized to represent you. This is not an optional procedure; this is the person you will be working with for emergency communications.
This is an annual cost, and it is a major factor in the cost of FDA registration in India.
- Consultancy Fees
For most Indian companies, it makes sense to hire a consultancy or law firm to help with the technical aspects. This is another major factor in the overall cost for FDA registration in India and includes:
- Application Preparation: Information collection, form completion, and ensuring that the Dun & Bradstreet number is correctly synchronized.
- Compliance Advice: Strategic advice for GMPs (Good Manufacturing Practices) and HACCP.
- Document Review: Ensuring that facility information and product lists meet stringent U.S. requirements.
- Ongoing Support: This is an annual subscription that also includes biennial renewal.
Indirect but Crucial Market Entry Cost
- Testing & Certification Costs
Your product, depending upon whether it is spices, honey, or dairy, may require certain tests to prove your compliance with US safety standards. Even though these costs are indirect, they are essential to pass customs clearance and avoid Import Alerts.
- Labelling Compliance
USFDA has very stringent requirements for ingredient listing, nutritional information, and allergen statements.
Incorrect labeling is one of the leading reasons for shipments being detained at US ports.
Label review and redesign can cost anywhere between ₹25,000 to ₹60,000 per product.
>Helpful Guide:- USFDA vs FSSAI: The Complete 2026 Breakdown — Jurisdiction, Cost, Process & Who Needs Both
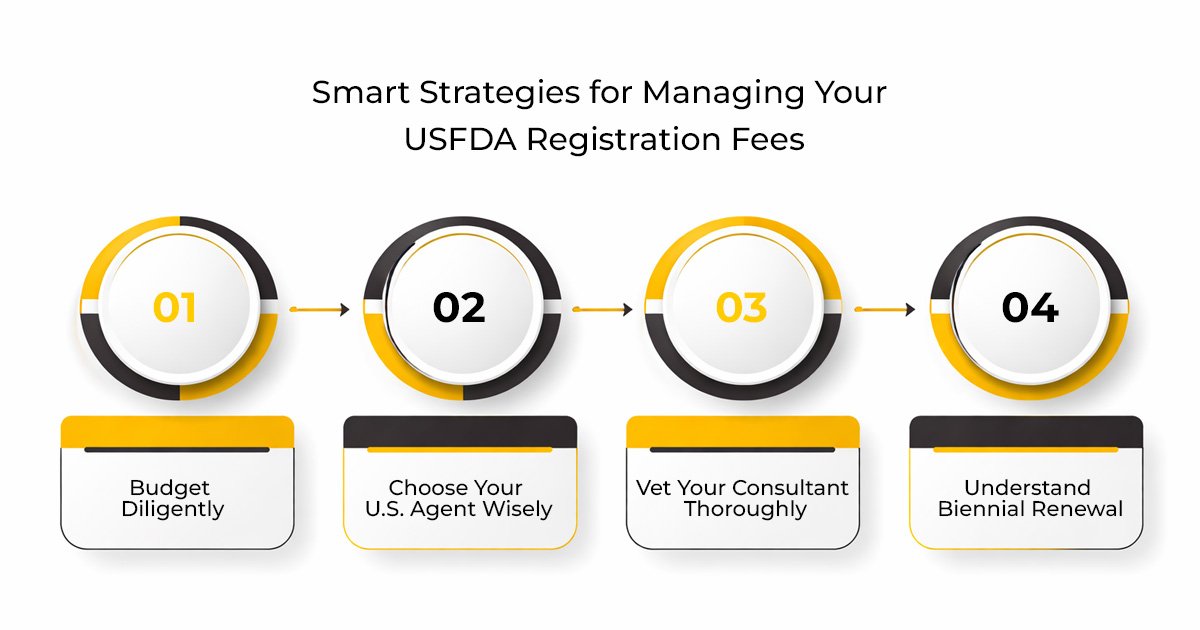
Smart Strategies for Managing Your USFDA Registration Fees
To effectively manage USFDA registration fees, it is important to have a planned approach towards managing the overall FDA registration cost India.
- Budget Diligently: A detailed budget should be planned, which includes not only consultancy charges and US Agent charges but also potential charges for lab testing, label charges, and annual charges.
- Choose Your U.S. Agent Wisely: A US Agent plays a very important role in this entire USFDA registration process. A US Agent is an essential part of USFDA registration fees India; hence, it is important to choose a US Agent wisely.
- Vet Your Consultant Thoroughly: Don’t look to save money on the FDA price India; look to save money on costly mistakes down the road. Choose a consultant who has expertise in USFDA regulations for Indian exporters, has a transparent fee structure, and has good client reviews.
- Understand Biennial Renewal: Mark your calendar to remember to renew between Oct 1 – Dec 31 of even-numbered years to avoid last-minute charges.
Guide to Managing USFDA Registration Fees
- However, it is possible to attempt it yourself, which is not recommended as it may lead to costly delays and/or denial of registration by USFDA. It is better to invest in a consultant.
- Several U.S. Agents or Consultants offer registration, U.S. Agent service, and biennial renewal in a package that will give you a better FDA price India.
- Well-documented facilities and information will minimize the cost and time that a consultant will have to spend in preparation of your application.
- Keep yourself updated by regularly visiting the FDA website for updates in regulations. Lack of such updates may lead to costly mistakes.
- Look at the USFDA registration fees as a strategic investment in a premium global market for your business.
>Top Trending Guide:- BRC Full Form, Meaning, Certification Process & Cost in India (2026 Guide)
Frequently Asked Questions
What are the USFDA registration fees for Indian exporters?
The USFDA itself does not charge any registration fee,costs majorly come from mandatory US agent fees and optional consultancy fees for application aid.
What is the FDA registration cost for food and pharma India?
There is no fees for food facility registration with the FDA, though there are significant user fees, such as PDUFA fees, paid directly to the FDA for pharma registration.
What are the USFDA license fees and charges India?
It is important to note that the term license fees is misleading, as there are no direct license fees from the USFDA for food facility registration.
How to export USA FDA food products from India?
You are required to register your food facility with the USFDA, appoint a U.S. Agent, ensure compliance of your products as well as good manufacturing practices (GMPs).
What is the USFDA export process for food items?
The process includes obtaining an FDA Registration Number, ensuring compliance of products and labels, and having a designated US Agent to help communicate with the FDA.
What are the FDA export requirements for Indian food manufacturers?
The key requirements include a registered food facility, compliance with CGMPs and a Hazard Analysis and Risk-Based Preventive Controls (HARPC) program
Is the USFDA fees india for registration a one-time payment or recurring?
Although registration is free, there is a required annual fee for a U.S. Agent service, and registration must be renewed every two years, making it a recurring expense.