For Indian manufacturers and Food Business Operators, while the opportunity to penetrate the vast and lucrative US market is an attractive one, it is often marred by the daunting task of complying with regulations. Trust and quality assurance are of utmost importance, especially when dealing with international regulations. The US Food and Drug Administration is at the helm of affairs, requiring strict compliance with its regulations before any product is allowed to enter its territory. While it may seem a daunting task, understanding the usfda registration process is your first step to unlocking unprecedented global opportunities. This is a detailed guide to demystify the fda registration steps to provide clarity to Indian Food Business Operators seeking to penetrate the lucrative US market.
 Need help with the USFDA Registration Process?
Need help with the USFDA Registration Process?
Legal4sure experts guide you through documentation, application filing, and compliance requirements to complete your USFDA registration smoothly and start exporting to the USA without delays.
Understanding USFDA Mandates
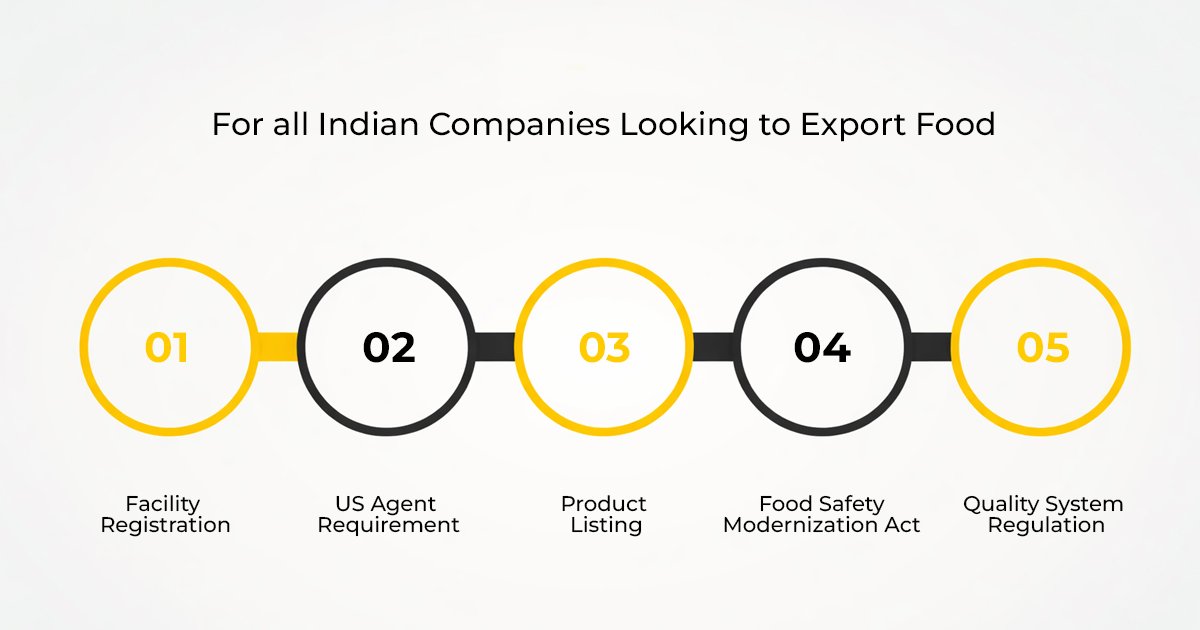
For all Indian companies looking to export food, drugs, and medical devices to the United States, it is not only optional but also mandatory to comply with the USFDA regulations. This is because these are legal and often buyer-specified guidelines that are set for the safety and efficacy of the products for the end-consumer.
Some of the major requirements are:
- Facility Registration: All domestic and foreign facilities that manufacture, process, pack, or hold food, drugs, and medical devices intended for consumption or use in the United States must register with the FDA. This facility registration must be renewed annually for food facilities between October 1 and December 31.
- US Agent Requirement: Foreign facilities must register with the FDA through a US Agent. This acts as a vital bridge for communication between the FDA and the foreign facility.
- Product Listing: Depending on the type of products (for example, medical devices and human drugs), there is often a need for the products to be listed with the FDA.
- Food Safety Modernization Act (FSMA) Compliance: Food facilities must comply with Food Safety Modernization Act regulations, including the development of Food Safety Plans (FSPs) similar to HACCP, and Foreign Supplier Verification Programs (FSVPs).
- Quality System Regulation (QSR): Medical device manufacturers must establish and maintain a quality system in compliance with 21 CFR 820.
>Helpful Guide:- What is USFDA Certification and Why It Matters
The USFDA Registration Process Step-by-Step
Compliance with USFDA can be an effortless process with proper planning. Below is the step-by-step process to follow while navigating the usdfa process india:
- Determine Applicability:
-
-
- Determine whether your facility is subject to FDA jurisdiction (e.g., food, dietary supplement, drug, medical devices, cosmetic).
- Understand the particular regulatory pathway for your product category.
-
- Assign a US Agent:
-
-
- This is a mandatory step for all foreign facilities.
- Your US Agent should be physically located in the United States and accessible for direct communication with the FDA on your behalf.
- Assign a US Agent because they will serve as your main point of contact for normal communications and emergency situations.
-
- Secure a DUNS Number (for Food Facilities):
-
-
- If you are registering a food facility, you will need to acquire a Data Universal Numbering System (DUNS) number from Dun & Bradstreet.
- Please ensure that your company details linked to your DUNS number are correct and current.
-
- Establish an FDA FURLS Account:
-
-
- Create an account on the FDA’s Unified Registration and Listing System (FURLS). FURLS is the portal through which all registrations and listings are conducted.
- managed.
-
- Complete Facility Registration:
-
-
- Give detailed information about your facility, including the name, address, contact details, ownership, and the operations carried out within the facility, such as manufacturing, processing, or packing.
- For food facilities, you are required to declare your compliance with the requirements of the FSMA. You are then required to give details about your food safety plan.
-
- Product Listing (if applicable):
-
-
- For medical devices and drugs, you are required to list the products you wish to export, including details about the products, their classification, and their use. This step may come after facility registration.
- For medical devices and drugs, you are required to list the products you wish to export, including details about the products, their classification, and their use. This step may come after facility registration.
-
- Submit and Receive Confirmation:
-
-
- Review all the information carefully to avoid any mistakes before submitting the details to the FDA.
- Once you have successfully submitted the details, you are issued a registration number, and you are sent a confirmation email. You are then required to keep this document safe.
-
- Annual Renewal:
-
- It is essential to note that FDA registrations, especially for food facilities, are required to be renewed annually. Make sure to keep this in mind and look for the renewal period, which is between October 1st and December 31st for food facilities.
- It is essential to note that FDA registrations, especially for food facilities, are required to be renewed annually. Make sure to keep this in mind and look for the renewal period, which is between October 1st and December 31st for food facilities.
>Helpful Guide:-How to Get US FDA Certification in India in a Simple Way
Struggles Of USFDA Registration Process
The problems that Indian businesses face with regard to the usfda process india usually lie in a few areas:
- Information Overload & Gap: With a huge number of FDA regulations to abide by, it is a daunting task to identify specific regulations that are applicable to a particular business and product.
- Lack of Specialized Knowledge: Indian SMEs usually lack in-house knowledge with regard to US regulations and find it difficult to interpret FDA regulations correctly.
- Resource Constraints: With limited resources available at hand, it is a daunting task to allocate resources to what appears to be a tedious and time-consuming international compliance process.
- Reactive vs. Proactive Approach: Instead of making it an integral part of the business from day one, businesses usually adopt a reactive approach to problems, which is not only costly but also troublesome.
Advantages of USFDA Compliance
Compliance with fda registration requirements is more than just compliance with the law; it is a strategic business move that offers many advantages over the competition:
- Increased Access to Markets: Directly unlocks access to the massive U.S. consumer market, thereby opening up new revenue streams.
- Increased Credibility and Trust: Signifies commitment to international quality, safety, and regulatory requirements, thereby generating immense trust among U.S. consumers, distributors, and end-users.
- Increased Business Efficiencies and Reduced Risk: Significantly reduces the risk of product detention, refusal, and costly regulatory fines at the U.S. border.
- Increased Competitive Advantage: Sets you apart from non-compliant competitors, thereby giving you a significant edge over them.
Insights for USFDA Success
- Think of the USFDA approval process as an opportunity to invest in global trust and market longevity.
- Selecting your US Agent is critical; they are your official point of contact and will facilitate timely communications with the FDA.
- Developing strong internal documentation and quality management systems is essential for streamlining the entire process.
- Preemptive planning and early adoption of regulatory requirements are critical for minimizing costly delays and even potential rejection of products.
- Don’t underestimate the importance of the annual renewals; failure to comply will cause immediate market access problems.
>Helpful Guide:- USFDA Certification Cost 2026 – Full Fee Breakdown & What You Must Know
Frequently Asked Questions
What is the usfda registration process step by step for exporters india?
It entails the selection of a US Agent, acquisition of a DUNS number, and the creation of an FDA account, followed by facility registration and the timely renewal of the account annually.
How to register food facility with usfda india?
The process entails the selection of a US Agent, acquiring of a DUNS number, and facility registration through the FDA’s FURLS system, all in compliance with FSMA regulations on food safety.
What is the fda registration procedure for indian manufacturers of medical devices?
Medical devices manufacturers from India must register their establishment and list their devices with the FDA, which entails pre market submissions such as 510(k) and PMA.
How long does the fda approval process typically take?
While facility registration is often a speedy process taking only weeks, pre-market submissions for devices and drug approvals can take months or even years.
What documents needed usfda?
Facility registration requires basic information such as facility details (name, address, and owner), contact details, product types, and the selection of a US Agent.
What are the fda requirements india for medical devices?
Device manufacturers are required to classify their device, develop a Quality Management System (QMS), list their device, and register their facility, often with a US Agent.
Do I need a usfda checklist?
A USFDA Checklist, although not mandatory, can prove to be a valuable tool in organizing your information and preparing you for audits.